8.Clostridioides difficile 初次感染,依其嚴重程度,下列處方何者最適當?
(A)nonsevere,metronidazole 250 mg PO Q8H
(B)severe,vancomycin 125 mg PO Q6H
(C)severe,fidaxomicin 200 mg PO Q8H
(D)fulminant with ileus,metronidazole 500 mg IV Q8H+vancomycin 250 mg rectal enema Q6H
統計: A(28), B(54), C(11), D(24), E(0) #3817523
詳解 (共 6 筆)
-
直接回答你:
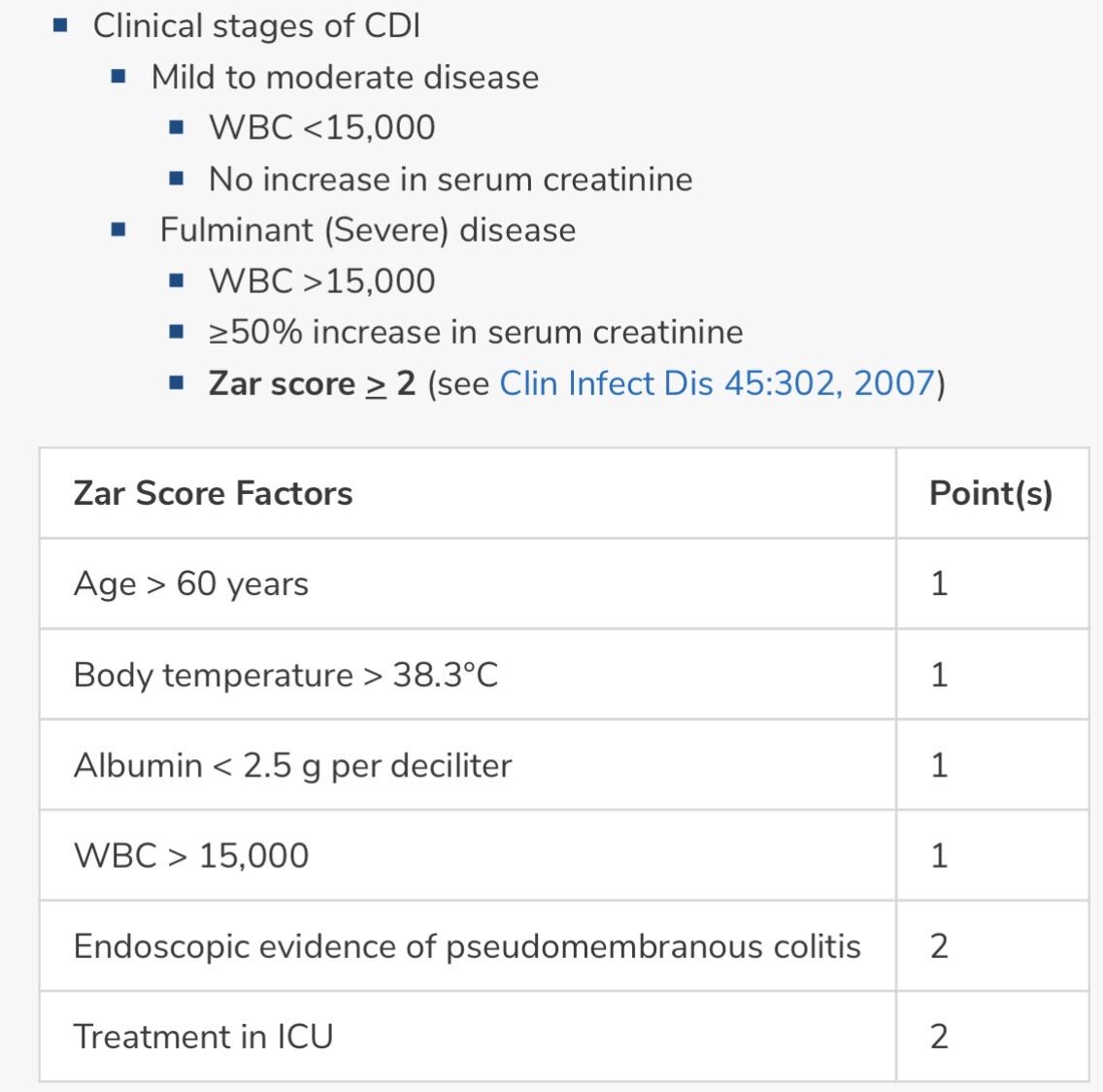
CDI (艱難梭菌感染) 總共分成三級:
-
輕症 (Non-severe)
-
重症 (Severe)
-
猛爆性 (Fulminant) $\leftarrow$ 只有這一級會有「腸阻塞 (Ileus)」!
❌ (A) nonsevere:metronidazole 250 mg PO Q8H
-
錯誤點 1 (藥物地位): 現在就算是輕症 (Non-severe),首選也是 Fidaxomicin 或 Vancomycin。Metronidazole 只是替代方案。
-
錯誤點 2 (劑量): 就算真的要用 Metronidazole,劑量標準是 500 mg PO TID (Q8H)。250 mg 劑量太低,治療失敗率高。
-
-
✅ (B) severe:vancomycin 125 mg PO Q6H
-
正確!
-
指引建議: 不論是輕症 (Non-severe) 還是重症 (Severe) 的初次感染,口服 Vancomycin 的標準劑量都是 125 mg QID (一天四次, Q6H) 吃 10 天。
-
這是一個非常標準且安全的答案。
-
-
❌ (C) severe:fidaxomicin 200 mg PO Q8H
-
錯誤點 (頻次): Fidaxomicin 是長效且很貴的藥。
-
正確用法: 200 mg PO BID (一天兩次, Q12H) 吃 10 天。不是 Q8H。
-
-
❌ (D) fulminant with ileus:metronidazole 500 mg IV Q8H + vancomycin 250 mg rectal enema Q6H
-
錯誤點 (缺角): 猛爆性 (Fulminant) 感染的治療核心是 「口服高劑量 Vancomycin」。
-
正確指引建議:
-
Vancomycin 500 mg PO/NG Q6H (一定要給高劑量口服,若腸阻塞則透過鼻胃管給)。<-- 選項漏了這個最重要的!
-
Metronidazole 500 mg IV Q8H (這部分選項有寫對)。
-
若有腸阻塞 (Ileus),加用 Vancomycin Rectal enema (灌腸),通常劑量是 500 mg in 100 ml NS Q6H (選項寫 250 mg 偏低,且最致命的是漏掉了口服 Vancomycin)。
-
-
Primary Regimens
- Treatment considerations
- If possible, stop the inciting antibiotic
- Up to 20-25% patients relapse though less frequent with fidaxomicin
- In general, avoid antiperistaltic medicine during acute phase
- Generally 10 days recommended, but course can be extended to 14 days if improving but not resolved.
- Mild Disease or Moderate disease, initial episode:
- Fidaxomicin 200 mg po bid x 10 days
- Vancomycin 125 mg po qid x 10 days
- Fulminant (Severe) disease, initial episode:
- Vancomycin 500 mg q6h po or via nasogastric tube ± Metronidazole 500 mg IV q8h, particularly if ileus is present
- For patients with ileus, administer Vancomycin 500 mg in 100 mL normal saline per rectum as a retention enema q6h.
- In retrospective study of ICU patients, Vancomycin + Metronidazole combination associated with decrease in mortality from 36 to 16%.
- Vancomycin 500 mg q6h po or via nasogastric tube ± Metronidazole 500 mg IV q8h, particularly if ileus is present
- Severe disease with toxic megacolon: treatment as above PLUS
- Sometimes only option is colectomy
- Alternative: loop ileostomy coupled with antegrade colonic irrigation with Vancomycin + intravenous Metronidazole (Ann Surg 254:423, 2011)
- No data on the efficacy of Fidaxomicin in patients with severe life-threatening disease.
- Fecal microbiota transplant (FMT) is promising; supportive evidence in one retrospective cohort study (Open Forum Infect Dis 6:ofz398, 2019)
- Sometimes only option is colectomy
- Recurrent CDI:
- First Recurrence (use a regimen different from original, if possible)
- Fidaxomicin 200 mg po bid x 10 days
- Fidaxomicin (extended regimen) 200 mg po bid x 5 days, 200 mg po qod x 20 days
- Vancomycin 125 mg po qid x 10 days
- Vancomycin taper as follows:
- 125 mg po qid x 10 days, followed by
- 125 mg tid x 1 week, then
- 125 mg bid x 1 week, then
- 125 mg q24h x 1 week, then
- 125 mg q48h x 1 week, then
- 125 mg q3w x 1 week
- Recurrence following treatment with Metronidazole as primary regimen
- Vancomycin 125 mg po qid x 10 days
- Fidaxomicin 200 mg po bid x 10 days
- Multiple recurrences:
- Fecal microbiota transplantation (FMT) emerging as a treatment of choice for recurrent infections.
- For details of donor screening used for commercial product from Open Biome see N Engl J Med 2019; 381:2070.
- Vancomycin 125 mg po qid x 10 days followed by Rifaximin 400 po tid x 20 days
- FMT is becoming more logistically difficult due to emergence MDR fecal contents, increased regulations (see comments)
- Commercial fecal microbiota products approved by FDA for recurrent episodes reviewed in Clin Infect Dis 77:S487 2023.
- Rebyota (rectally-administered, live fecal microbiota suspension): Per rectum as single dose enema. No need for prior bowel prep. Treatment success at 8 weeks: 75%. See Med Lett Drugs Ther 65:35, 2023
- Vowst (oral fecal microbiota spore capsules): Give 10 oz magnsesium citrate. After 24 hr, 4 capsules po daily for 3 consecutive days. Treatment success at 8 weeks: 83%. See Med Lett Drugs Ther. 65:81, 2023
- First Recurrence (use a regimen different from original, if possible)
- Prophylaxis to prevent recurrent CDI in patients requiring on-going systemic antimicrobial therapy for another indication:
- Retrospective study of treatment doses of oral Vancomycin while patient was receiving systemic antibiotics found reduced incidence of recurrent CDI to 4.2% versus 26.6% in patients not treated with concurrent vancomycin (Clin Infect Dis 63:651, 2016).
- Prevention of C. difficile in allogeneic hematopoietic stem cell recipients with oral vancomycin prophylaxis. Compared frequency of C. diff. over 2 sequential periods. CDI occurred in 31 of 131 (23.6% in the control group and 1 of 71 of those given vancomycin prophylaxis